- PATIENT FORMS | REQUEST A CONSULTATION | CONTACT US
- 1-844-NSPC-DOC
Young college student with severe headache and a large intracerebral bleed
Minimally Invasive Approach for Spinal Revision Surgery
October 26, 2021
74 Year Old Woman with Occipital Headaches
October 27, 2021Educational Goals:
Learners will be able to recognize the symptoms that may suggest Brain Arteriovenous Malformations, and order appropriate tests to confirm the diagnosis, and refer these patients to a subspecialist for management and treatment.

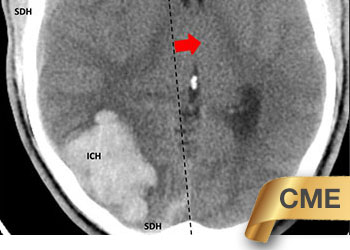
Fig 1. Head CT scan showing large right-sided, parieto-occipital intra-cerebral hemorrhage (ICH) and small occipital and temporal subdural hemorrhage (SDH) with right to left midline shift (red arrow).
While on her computer, a young female college student developed a severe headache. Having lived with migraines, she was initially not concerned. However, the headache worsened over the next few hours. She became progressively drowsy and was taken to the emergency room.
A head CT revealed a large posterior-parietal intracerebral bleed, an acute subdural clot, and right to left midline shift (Fig 1). Following the review of a head CTA, the patient was diagnosed with a ruptured brain arteriovenous malformation (AVM). Urgent treatment was indicated for this acute intracerebral hemorrhage (ICH) with significant mass effect and the risk of catastrophic rebleeding of the AVM.
On her initial exam, she was arousable to tactile stimulation, had a left visual field cut, mild upper extremity weakness, and some neglect.
The patient was taken to the angio-suite where an angiogram confirmed a high flow, high pressure AVM fed primarily by the posterior cerebral artery with a small collateral from the middle cerebral artery (Fig 2 and 3). The AVM drained into a tortuous, partially occluded and stenotic single draining vein.

Fig 2. AP view, mid-arterial phase of a right internal carotid artery (ICA) injection. The large tortuous draining vein (yellow star) with the suggestion of clot (dotted circle) can be seen. AVM nidus (red arrow), MCA (middle cerebral artery, ACA (anterior cerebral artery).

Fig 3. Lateral view, late-arterial phase of a right internal carotid artery (ICA) injection. Filling of the AMV (red arrow) by the fetal posterior cerebral artery (PCA) and small middle cerebral artery feeder (blue arrow). The large tortuous draining vein (yellow star) with venous stenosis and the suggestion of clot (dotted circle) can be seen.
The student underwent NBCA-embolization of the AVM via the dominant feeding posterior cerebral artery with care to avoid embolizing the calcarine branches and avoid NBCA penetration into the single draining vein (Fig 4 and 5). Following the intervention, a tiny feeder from the middle cerebral artery remained (Fig 4).
With this embolization, the acute risk of rebleeding was addressed and the risk of possible acute decompressive surgery reduced, should the mass effect become uncontrollable. In the future, the patient could continue the treatment electively after she had recovered from this intracerebral bleed.

Fig 4. Post-embolization lateral view (4A) and AP view (4B), late-arterial phase of a right internal carotid artery (ICA) injection. With near total obliteration of the AVM, a small MCA component (red arrow) can be seen working its way to the residual AVM (embolization material; yellow arrow). Residual veins (blue arrows) and spared calcarine branches of the posterior cerebral artery (CBr).

Fig 5. Comparison, pre (5A) and post-embolization (5B) AP view, late-arterial phase of a right internal carotid artery (ICA) injection. On the right, near total obliteration of the AVM (red arrow). Draining vein (yellow star), residual veins (blue stars).
Post-Procedural Clinical Course
The next morning, the patient was awake. Interestingly, control head CT showed partial resolution of the subdural hemorrhage with reduced mass effect. It was determined that when the blood clot had resorbed, the patient could be treated effectively with additional embolization, radio surgery, or direct surgery. After multiple consultations, the advantage of surgery was chosen by the patient. In her case, it was thought to have the greatest likelihood of immediate and complete cure, with the least follow-up burden.
The pre-surgical angiogram did, as expected, show some growth of the middle cerebral artery component, but no recanalization of the posterior cerebral artery component. Surgery was uneventful, leading to full obliteration of the AVM.
Follow-up visual field testing found complete resolution of the left hemianopia. The young patient is now back in college, neurologically normal, having had a busy and interesting summer.
Treatment Considerations
Together with initial intracranial hemorrhage, migraines and headaches are among the most common clinical events in the course of the natural history of brain AVMs1. Approximately 50% of patient with AVM’s have headaches. But headaches, even migraines are very common, with 10-20 % of the general population suffering from migraines. Other forms of presentation include seizure and focal neurological deficits. In the modern era of frequent use of brain imaging, the detection rate of brain AVMs is estimated to be approximately 1-1.5/100,000 patient years with an incidence of initial hemorrhage of approximately 0.5/100,000 patient years.
Studies have shown, like in this case, brain AVM bleeds can be dramatic. Although they can, they usually do not cause major disability on the first bleeding event2,3 which contrasts the outcomes following other causes of intracranial hemorrhage, such as hypertensive intracerebral bleeds or aneurysmal subarachnoid hemorrhages4. Brain AVMs can be located anywhere in the brain and cause any type of intracranial hemorrhage.
Emergency treatment is indicated when the rebleeding risk is considered high, e.g. due to the presence of intranidal aneurysms, venous occlusion with associated venous hypertension, or mass effect5,6. The acute rebleeding rate in cases not having these characteristics is quite low. However, hemorrhagic presentation is thought to be associated with bleeding events, i.e. increased rebleeding risk6.
The goal of treatment is total obliteration of the brain AVM to eliminate the risk of future bleeds. Treatment is often a multi-disciplinary approach, including endovascular embolization, surgical resection and radiation surgery. Complex brain AVMs may require multiples stages of treatment.
In addition to emergency treatment to reduce the risk of rebleeding, a long-term outlook is necessary with consideration of combined therapy7 and best timing of treatment, tailoring the management for the patient’s lifetime needs. Also, following the post-rupture emergent care, allowing the patient (and his/her brain) to recover before performing additional interventions is usually prudent.
So, what to do with unruptured or unbled brain AVMs? The only randomized control clinical trial for Unruptured Brain AVM’s, the NIH-sponsored ARUBA trial, was stopped early by the Data Safety Monitoring Board due to the overwhelming morbidity and mortality in the interventional/surgical arm compared to the medical arm8. The results highly suggest a primarily observational and non-invasive medical approach in cases of unruptured brain AVMs9.
References
- Choi JH, Mohr JP. Brain arteriovenous malformations in adults. Lancet Neurology 2005; 4: 299¬–308.
- van Beijnum J, Lovelock CE, Cordonnier C, Rothwell PM, Klijn CJM, Al-Shahi R, and the SIVMS Steering Committee and the Oxford Vascular Study. Outcome after spontaneous and arteriovenous malformation-related intracerebral haemorrhage: population-based studies. Brain 2009; 132: 537–43.
- Choi JH, Mast H, Sciacca RR, Hartmann A, Khaw AV, Mohr JP, Sacco RL, Stapf C. Clinical outcome after first and recurrent hemorrhage in patients with untreated brain arteriovenous malformation. Stroke 2006; 37: 1243–47.
- Stapf C, Mast H, Sciacca RR, Berenstein A, Nelson PK, Gobin YP, Pile-Spellman J, Mohr JP, and the New York Islands AVM Study Collaborators. The New York Islands AVM Study: design, study progress, and initial results. Stroke 2003; 34: e29–33.
- Choi JH, Pile-Spellman J. Pathophysiology of arteriovenous anomaly-related hemorrhage. In: Stroke Revisited: Hemorrhagic Stroke. Editor: Seung-Hoon Lee. Springer 2018.
- Stapf C, Mast H, Sciacca RR, Choi JH, Khaw AV, Connolly ES, Pile-Spellman J, Mohr JP. Predictors of hemorrhage in patients with untreated brain arteriovenous malformation. Neurology 2006; 66: 1350–55.
- Ogilvy CS, Stieg PE, Awad I, Brown Jr RD, Kondziolka D, Rosenwasser R, Young WL, Hademenos G. Recommendations for the management of intracranial arteriovenous malformations. Stroke 2001; 32: 1458-71.
- Mohr JP, Parides MK, Stapf C, and international ARIBA investigators. Medical management with or without interventional therapy for unruptured brain arteriovenous malformations (ARUBA): a multicentre, non-blinded, randomised trial. Lancet 2014; 383: 614-21.
- Mohr JP, Overbey JR, von Kummer R, and international ARUBA investigators. Functional impairment for outcomes in a randomized trial of unruptured brain AVMs. Neurology 2017; 89: 1499-1506.
Disclosures:
The planners and faculty participants do not have any financial arrangements or affiliations with any commercial entities whose products, research or services may be discussed in these materials.
CME Accreditation:
This activity has been planned and implemented in accordance with the accreditation requirements and Policies of the Medical Society of the State of New York (MSSNY) through the joint providership of the Academy of Medicine of Queens County and NSPC Brain and Spine Surgery. The Academy of Medicine of Queens County is accredited by The Medical Society of the State of New York (MSSNY) to provide continuing medical education for physicians. The Academy of Medicine of Queens County designates this Enduring Materials for a maximum of 1 AMA PRA Category 1 Credits™ as specified in this activity. Physicians should claim only the credit commensurate with the extent of their participation in the activity.